Hoe werkt OncoAct?
Van biopt tot rapport
Wat gebeurt er met je tumorweefsel en bloed als er een complete DNA-test op wordt uitgevoerd? Wat doet die complexe apparatuur precies? Hoe kom je erachter welke medicijnen voor jou zouden kunnen werken? Bekijk de video!
Whole genome sequencing

OncoAct maakt gebruik van whole genome sequencing waarmee het mogelijk is om het complete DNA van het tumorweefsel en van het bloed af te lezen.
Het menselijke DNA, ook wel genoom genoemd, bestaat uit 2 x 3 miljard ‘letters’ (één kopie van je moeder en één van je vader) en is aanwezig in iedere lichaamscel. Dit DNA bevat alle informatie die nodig is voor een cel, weefsel en organisme om te kunnen functioneren en voor cellen om te delen.
Door toeval en door omgevingsfactoren kan het DNA beschadigd raken. Hierdoor kan een cel andere eigenschappen krijgen en kan kanker ontstaan. Voor het goed begrijpen en optimaal kunnen behandelen van een tumor is het dus belangrijk om een hoge resolutie ‘foto’ van het beschadigde DNA uit de tumor te hebben.
DNA-beschadigingen komen voor in allerlei vormen. Het kan zijn dat enkele DNA-letters veranderd zijn, maar ook dat kleine of grote stukken DNA verdwenen of verdubbeld zijn. DNA kan ook breken en op de verkeerde manier gerepareerd worden door het lichaam, waarna het op een andere plek in het genoom terecht komt.
Met whole genome sequencing kunnen alle type veranderingen in kaart worden gebracht. Om zeker te weten dat de gemeten veranderingen mutaties zijn die alleen in de tumor voorkomen, wordt het DNA van de tumor altijd vergeleken met het ‘normale’ DNA van de patiënt. Voor dit laatste gebruiken we het DNA uit het bloed.
OncoAct WGS-rapport
Na een complete bioinformatische data-analyse kijken we welke bestaande en experimentele behandelingen aansluiten bij de eigenschappen (biomarkers) van de tumor, zowel voor geregistreerde indicaties als voor off-label indicaties. Hiervoor maken we gebruik van wereldwijde kennis verzameld in gespecialiseerde databases en overzichten van lopende onderzoeken in Nederland.
Alle relevante moleculaire gegevens en therapie opties worden samengevat in het OncoAct WGS-rapport. Hiermee heeft de behandelend arts een zo compleet mogelijk overzicht van de kankergerelateerde DNA-afwijkingen van de patiënt.
Het OncoAct WGS-rapport bevat de volgende gedetailleerde informatie:
- algemene samenvatting met relevant klinisch bewijs
- beschikbare medicijnen die mogelijk aansluiten bij de gevonden DNA-afwijkingen
- complete moleculaire details ter ondersteuning van een behandelkeuze
Verder geeft het patiëntrapport inzicht in:
- tumorspecifieke mutaties, amplificaties, deleties
- mutational load / mutational burden
- microsatelliet instabiliteit
- aanwezigheid van fusiegenen als gevolg van translocaties
- mogelijkheid voor deelname aan klinische studies in Nederland
De samenvatting bevat een overzicht van genen met potentieel ‘actionable mutations’ en diverse algemene tumorkarakteristieken, zoals microsatellite instability (MSI) en tumor mutational load (TML). Deze karakteristieken zijn belangrijk voor immunotherapie opties.
Klik hier voor een voorbeeld van het OncoAct WGS-rapport.
Wat vindt u in het OncoAct WGS-rapport?
De resultaten van alle bestaande DNA-testen in één rapport


Wat vindt u in het OncoAct WGS-rapport?
Een samenvatting van de uitkomsten die relevant zijn voor een mogelijke behandeling
Wat vindt u in het OncoAct WGS-rapport?
Een uitgebreide weergave van alle veranderingen in het erfelijk materiaal (mutaties)
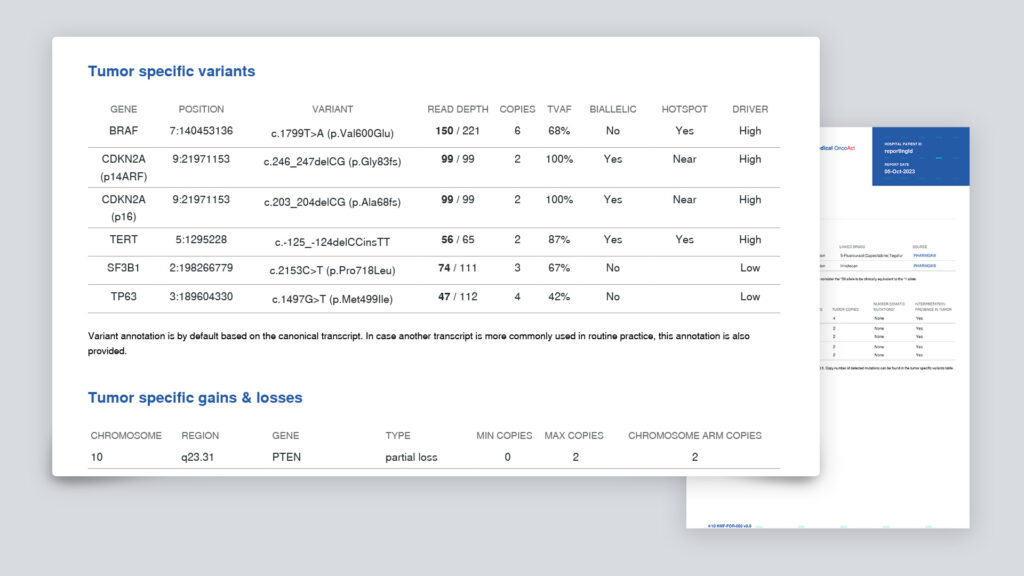

Wat vindt u in het OncoAct WGS-rapport?
Uitkomsten die altijd getoetst zijn aan de meest recente wetenschappelijke inzichten
Hoe helpt het u?
Het rapport ondersteunt de keuze voor behandeling
- reguliere behandelingen
- beschikbare experimentele studies
- off-label medicatie

Voordelen van een DNA-test op basis van whole genome sequencing
Alle mogelijke biomarkers
Alle mogelijke DNA-gebaseerde biomarkers worden in één DNA-test bepaald
Eén rapport
Alle resultaten en uitslagen worden in één rapport samengevat
Eén overzicht
Relevante resultaten met mogelijke behandelopties worden in één overzicht samengevat
Patiënten geanalyseerd
Gedurende de afgelopen jaren hebben we meer dan 5.100 patiënten met uitgezaaide kanker geanalyseerd in studieverband met whole genome sequencing.
Het aantal voor behandeling mogelijk relevante bevindingen verschilt sterk tussen kankertypes.
Aanknopingspunten voor bestaand medicijn
Gemiddeld vinden we bij 31% van de patiënten aanknopingspunten voor behandeling met een bestaand medicijn.
Off-Label
Bij 13% van de patiënten betreft het een medicijn dat eigenlijk voor een ander tumortype is geregistreerd (off-label)*
* gepubliceerd in Nature, november 2019
Experimentele medicijnen
Bij ongeveer 31% van de patiënten worden ook indicaties gevonden voor experimentele medicijnen. Toegang tot deze medicijnen is mogelijk via klinische studies.
Off-label behandeling
Een recente studie laat zien dat 34% van de patiënten baat heeft bij off-label behandeling in klinisch studieverband.*
* gepubliceerd in Nature, september 2019
Patiëntrapport
Behandeladvies naar aanleiding van complete DNA-analyses bij patiënten in de afgelopen 4 jaar
Doorlooptijd
Het is voor de patiënt en voor de behandelend arts belangrijk om snel te weten wat de uitslag is van OncoAct.
Over het algemeen sturen wij een patiëntrapport 10 tot 15 werkdagen na ontvangst van het tumorweefsel en bloed aan de behandelend arts.
Werkzaamheden tijdens de DNA-test
Het tumorweefsel en bloed worden na ontvangst en registratie direct ingepland voor verwerking. Het verwerken van het tumorweefsel en bloed bestaat uit een aantal bewerkelijke stappen, die elk één of meerdere dagen tijd kosten: DNA-isolatie, sample preparatie, DNA-sequencing en bioinformatische data-analyse. Na elke stap voeren wij kwaliteitscontroles uit. Wanneer het tumorweefsel voldoende tumorcellen bevat en het DNA van voldoende kwaliteit is voeren wij de test uit en kunnen wij de uitkomsten rapporteren. Indien dit niet het geval is, stellen wij geen OncoAct WGS-rapport op.
Validatie van de DNA-test
OncoAct wordt uitgevoerd conform gestandaardiseerde procedures in het ISO17025 geaccrediteerde laboratorium van Hartwig Medical Foundation in Amsterdam. Klik hier voor de scope van de accreditatie. De gebruikte methodes en testuitslagen zijn uitgebreid gevalideerd. U kunt deze per mail aanvragen.